(They can only be created by scientists in a laboratory.) Even today, researchers are still discovering new states of matter. 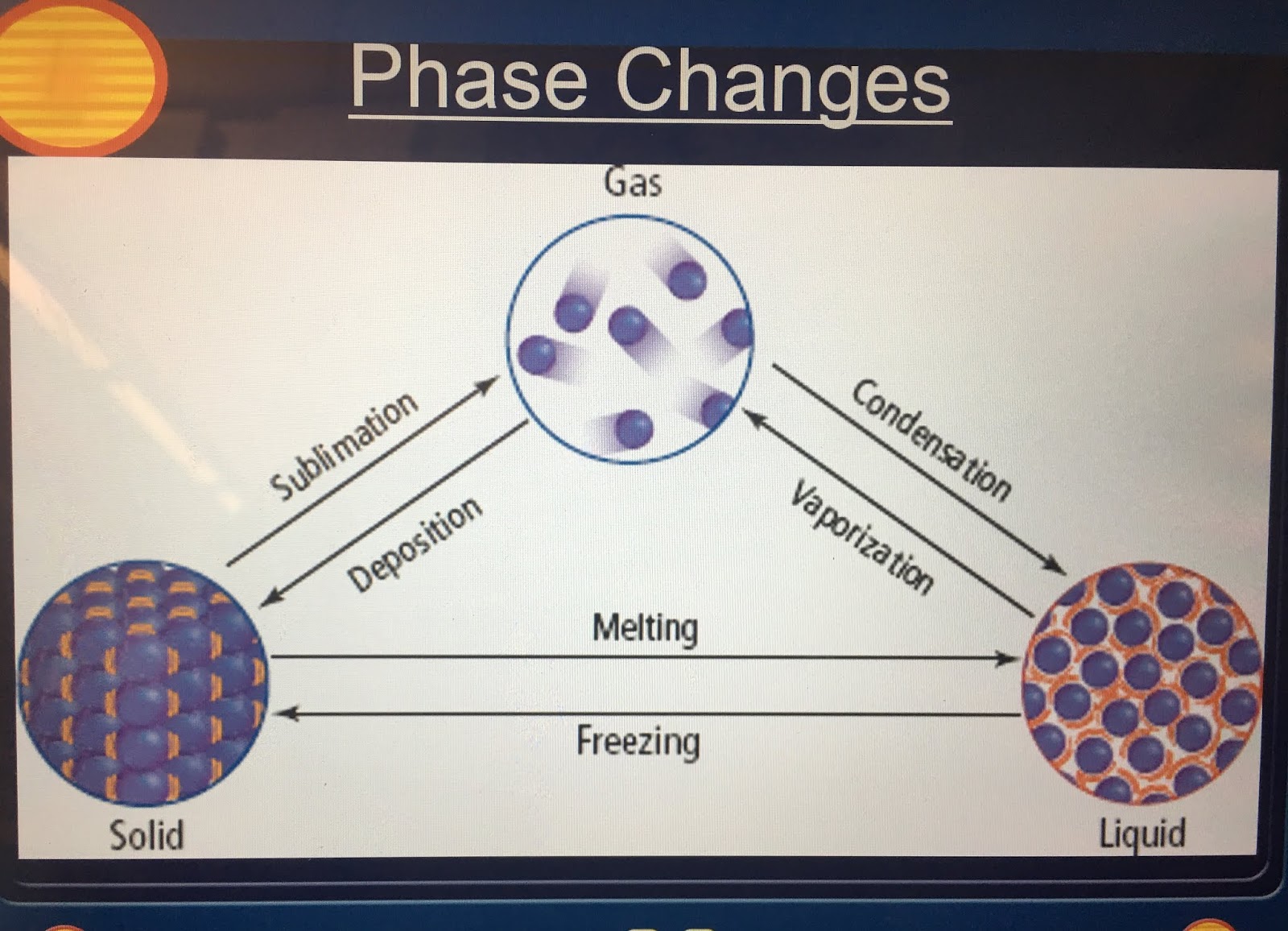
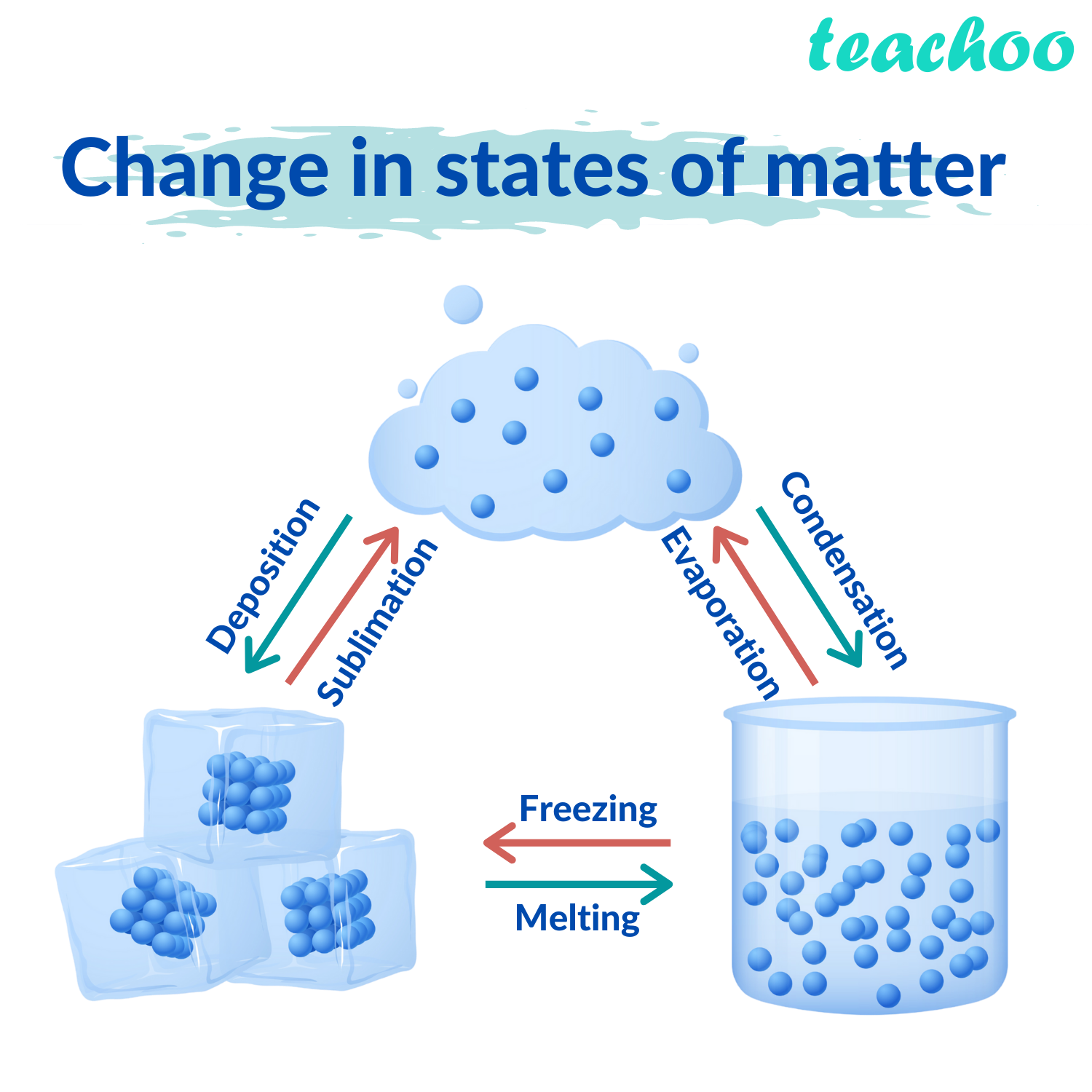
Lesser-known states develop under more extreme conditions - some of which never exist naturally on Earth. Solid, liquid and gas are the best-known states of matter. jack0m/DigitalVision Vectors/Getty Images Plus The arrows show the name of the process that moves each state of matter into another state. This diagram illustrates the cycle of the states of matter using H2O as an example. When these tiny building blocks of matter change their structure, their state or phase does too. All matter is made of atoms and/or molecules. However, water isn’t the only type of matter that changes states as it’s heated, cooled or compressed. In ancient Greece, one philosopher recognized how water could change form and reasoned that everything must be made of water. Solid (the ice), liquid (the water) and gas (the vapor) are the three most common states of matter - at least on Earth. 
This vapor is still H 2O, just in gas form. If it gets hot enough, you will notice steam rising off of the liquid. Put the ice into a pot over a flame on the stove and it will melt back down to liquid. The substance in the tray is still the same chemical - H 2O only its state has changed. Pour water into the tray, stick it in the freezer and a few hours later that liquid water will have transformed into solid ice. Like other substances, water can take different forms as its surrounding environment changes. Ice, water and vapor are three distinctly different forms - or states - of water.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
